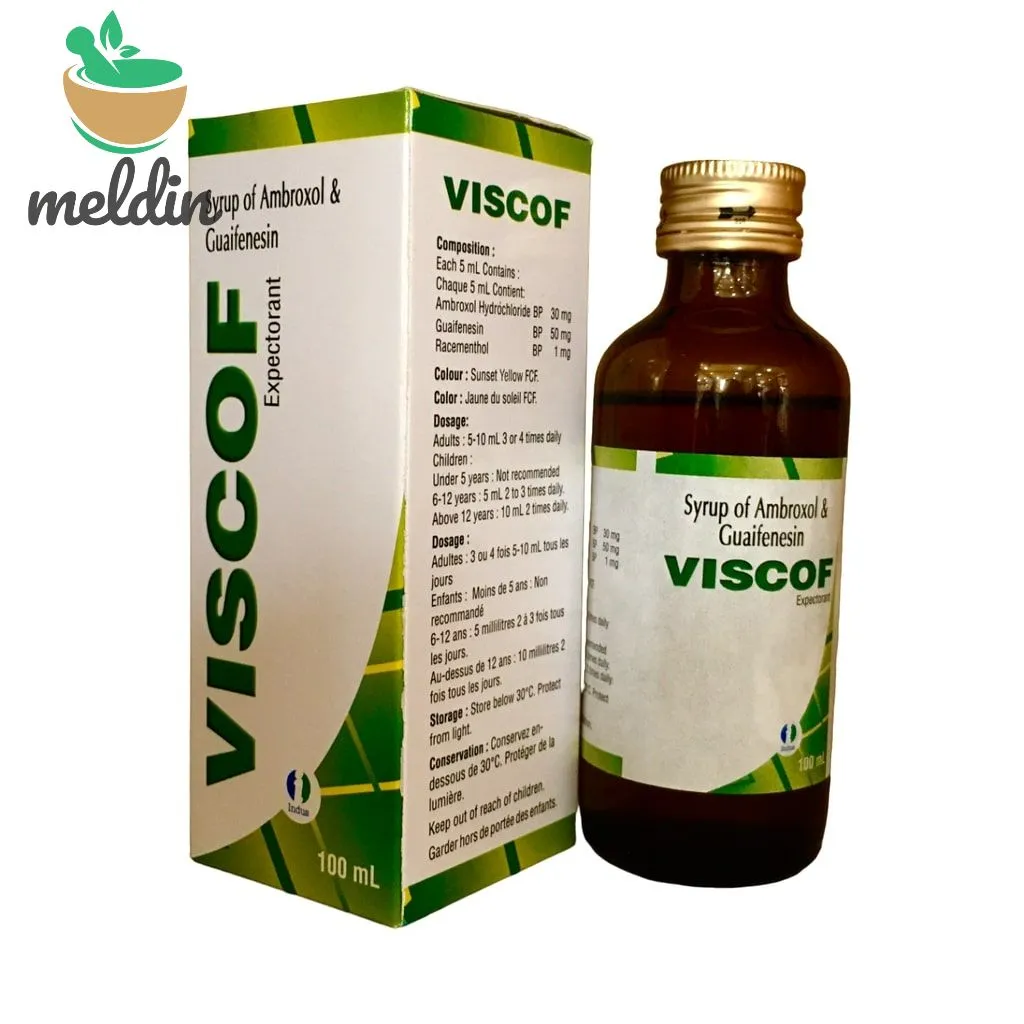
Viscof Syr
37.27 GH¢
37.27 GH¢
-
Brand
-
Volume
-
Variant
-
Marketer(s)
-
Manufacturer(s)
-
Active Ingredient(s)
-
Active Ingredient(s)
-
Active Ingredient(s)
-
Active Ingredient(s)
-
Active Ingredient(s)
-
Active Ingredient(s)
-
Active Ingredient(s)
-
Medicine Schedule
-
Therapeutic Classification
-
Therapeutic Classification
-
Therapeutic Classification
-
Route of Administration
-
Route of Administration
-
Pack Size
-
FDA Reg No:
-
FDA Reg No:
-
FDA Reg No:
-
Packaging
| Variant | Expectorant, D, S |
| Brand | Indus |
| Volume | 100ml |
| Marketer(s) | Indus Life Sciences Pvt Ltd |
| Manufacturer(s) | Sai Mirra Innopharm Pvt Ltd |
| Active Ingredient(s) | Ambroxol, Guaifenesin, Menthol, Salbutamol Sulfate, Dextromethorphan Hydrobromide, Phenylephrine Hydrochloride, Cetrizine Hydrochloride |
| Medicine Schedule | Over-the-Counter (OTC) |
| Therapeutic Classification | R – Respiratory System, R03 Drugs For Obstructive Airway Diseases, R05 Cough And Cold Preparations |
| Route of Administration | Oral (Enteral), Syrup |
| Pack Size | Ctn of 25 |
| FDA Reg No: | N/A, N/A, N/A |
| Brand: Indus |
| Volume: 100ml |
| Marketer(s): Indus Life Sciences Pvt Ltd |
| Manufacturer(s): Sai Mirra Innopharm Pvt Ltd |
| Active Ingredient(s): Ambroxol , Guaifenesin , Menthol , Salbutamol Sulfate , Dextromethorphan Hydrobromide , Phenylephrine Hydrochloride , Cetrizine Hydrochloride |
| Medicine Schedule: Over-the-Counter (OTC) |
| Therapeutic Classification: R – Respiratory System , R03 Drugs For Obstructive Airway Diseases , R05 Cough And Cold Preparations |
| Route of Administration: Oral (Enteral) , Syrup |
| Pack Size: Ctn of 25 |
| FDA Reg No:: N/A , N/A , N/A |