Dithranol Oint
21.41 GH¢
21.41 GH¢
-
Brand
-
Manufacturer(s)
-
Dosage Unit
-
Weight
-
Active Ingredient(s)
-
Medicine Schedule
-
Therapeutic Classification
-
Therapeutic Classification
-
Route of Administration
-
Route of Administration
-
Pack Size
-
FDA Reg No:
-
Packaging
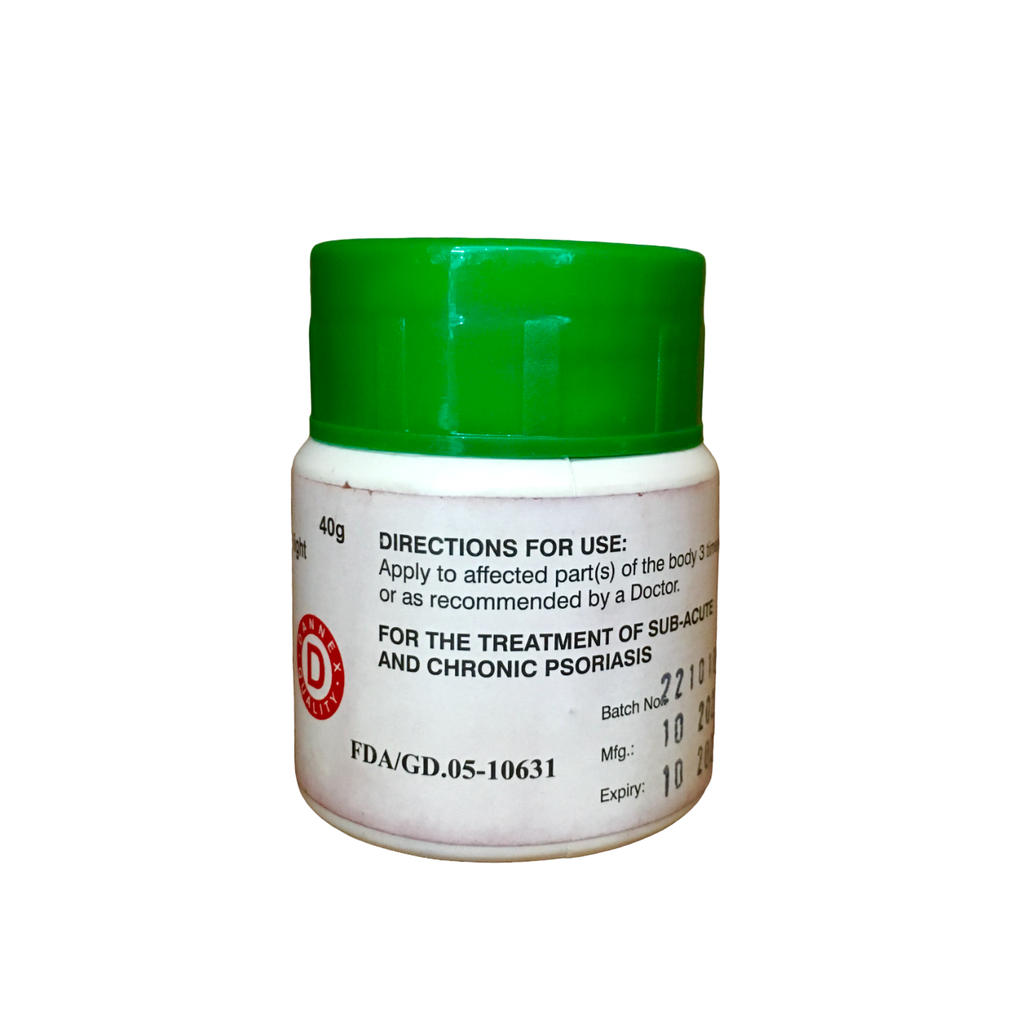
| Brand | Dannex |
| Manufacturer(s) | Dannex Ayrton Starwins |
| Dosage Unit | 0.1% w/w |
| Weight | 40gm |
| Active Ingredient(s) | Dithranol |
| Medicine Schedule | Over-the-Counter (OTC) |
| Therapeutic Classification | D – Dermatologicals, D05 Antipsoriatics |
| Route of Administration | Topical (Skin & Local Application), Ointment |
| Pack Size | Ctn of 120 |
| FDA Reg No: | FDA/GD.05-10631 |
| Brand: Dannex |
| Manufacturer(s): Dannex Ayrton Starwins |
| Dosage Unit: 0.1% w/w |
| Weight: 40gm |
| Active Ingredient(s): Dithranol |
| Medicine Schedule: Over-the-Counter (OTC) |
| Therapeutic Classification: D – Dermatologicals , D05 Antipsoriatics |
| Route of Administration: Topical (Skin & Local Application) , Ointment |
| Pack Size: Ctn of 120 |
| FDA Reg No:: FDA/GD.05-10631 |